FLAREX®
(fluorometholone acetate ophthalmic suspension) 0.1%
More Information
Indications and Usage
FLAREX® (fluorometholone acetate ophthalmic suspension) 0.1% is indicated for use in the treatment of steroid-responsive inflammatory conditions of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the eye.
Important Safety information
CONTRAINDICATIONS
Contraindicated in acute superficial herpes simplex keratitis, vaccinia, varicella, and most other viral diseases of the cornea and conjunctiva; mycobacterial infection of the eye; fungal diseases; acute purulent untreated infections, which like other diseases caused by microorganisms, may be masked or enhanced by the presence of the steroid; and in those persons who have known hypersensitivity to any component of this preparation.
WARNINGS AND PRECAUTIONS
Topical Ophthalmic Use: Not for injection.
Intraocular Pressure Increase: Prolonged use may result in glaucoma, damage to the optic nerve, and defects in visual acuity and visual field. It is advisable that the intraocular pressure be checked frequently.
Cataracts: Use of corticosteroids may result in cataract formation.
Delayed Healing: Topical ophthalmic corticosteroids may slow corneal wound healing. In those diseases causing thinning of the cornea or sclera, perforation has been known to occur with chronic use of topical steroids.
Viral Infections: Use in the treatment of herpes simplex infection requires great caution.
Bacterial Infections: Use of corticosteroids may suppress the host response and thus aid in the establishment of secondary ocular infections. Acute purulent infections of the eye may be masked or exacerbated by the presence of steroid medication.
Fungal Infections: Fungal infections of the cornea are particularly prone to develop coincidentally with long-term local steroid application. Fungus invasion must be considered in any persistent corneal ulceration where a steroid has been used or is in use.
Contamination: Do not touch dropper tip to any surface as this may contaminate the suspension.
Contact Lens Wear: Contact lenses should be removed during instillation of FLAREX but may be reinserted 15 minutes after instillation.
Temporarily Blurred Vision: Vision may be temporarily blurred following dosing with FLAREX. Care should be exercised in operating machinery or driving a motor vehicle.
ADVERSE REACTIONS
Glaucoma with optic nerve damage, visual acuity and field defects, cataract formation, secondary ocular infection following suppression of host response, and perforation of the globe may occur. Post marketing Experience: The following reaction has been identified during post marketing use of FLAREX in clinical practice. Because reactions are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The reaction, which has been chosen for inclusion due to either its seriousness, frequency of reporting, possible causal connection to FLAREX, or a combination of these factors, includes dysgeusia. The following rare adverse reactions have been reported: Cushing’s syndrome and adrenal suppression may occur after very frequent use of topical ophthalmic corticosteroids, particularly in very young children.


FRESHKOTE®
FRESHKOTE® Preservative Free (PF) is a lubricant eye drop that temporarily relieves burning, itching and other dry eye symptoms.

IHEEZO®
(chloroprocaine HCl ophthalmic gel) 3%
More Information
Indications and Usage
IHEEZO® (chloroprocaine HCl ophthalmic gel) 3% is indicated for ocular surface anesthesia.
Important Safety information
IHEEZO is contraindicated in patients with a history of hypersensitivity to any component of this preparation.
IHEEZO should not be injected or intraocularly administered.
Patients should not touch the eye for at least 10 to 20 minutes after using anesthetic as accidental injuries can occur due to insensitivity of the eye.
Prolonged use of a topical ocular anesthetic may produce permanent corneal opacification and ulceration with accompanying visual loss.
Do not touch the dropper tip to any surface as this may contaminate the gel.
IHEEZO is indicated for administration under the direct supervision of a healthcare provider. IHEEZO is not intended for patient self-administration.
The most common adverse reactions in studies following IHEEZO administration (incidence greater than or equal to 5%) were mydriasis, conjunctival hyperemia, and eye irritation.
You are encouraged to report suspected adverse reactions to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.


More Information
IOPIDINE®
(apraclonidine hydrochloride ophthalmic solution) 1% as base
More Information
Indications and Usage
IOPIDINE 1% Ophthalmic Solution is indicated to control or prevent post-surgical elevations in IOP that occur in patients after argon laser trabeculoplasty, argon laser iridotomy or Nd:YAG posterior capsulotomy.
Important Safety information
CONTRAINDICATIONS
IOPIDINE 1% Ophthalmic Solution is contraindicated for patients receiving monoamine oxidase inhibitor therapy and for patients with hypersensitivity to any component of this medication or to clonidine
WARNINGS AND PRECAUTIONS
Since IOPIDINE 1% Ophthalmic Solution is a potent depressor of IOP, patients who develop exaggerated reductions in IOP should be closely monitored. Although the acute administration of two drops of IOPIDINE 1% Ophthalmic Solution has minimal effect on heart rate or blood pressure in clinical studies evaluating patients undergoing anterior segment laser surgery, the preclinical pharmacologic profile of this drug suggests that caution should be observed in treating patients with severe cardiovascular disease including hypertension. IOPIDINE 1% Ophthalmic Solution should also be used with caution in patients with severe coronary insufficiency, recent myocardial infarction, cerebrovascular disease, chronic renal failure, Raynaud’s disease or thromboangiitis obliterans. The possibility of a vasovagal attack occurring during laser surgery should be considered and caution used in patients with history of such episodes. Topical ocular administration of two drops of 0.5%, 1%, and 1.5% IOPIDINE Ophthalmic Solution to New Zealand Albino rabbits three times daily for one month resulted in sporadic and transient instances of minimal corneal cloudiness in the 1.5% group only. No histopathological changes were noted in those eyes. No adverse ocular effects were observed in cynomolgus monkeys treated with two drops of 1.5% IOPIDINE Ophthalmic Solution applied three times daily for three months. No corneal changes were observed in 320 humans given at least one dose of IOPIDINE 1% Ophthalmic Solution.
ADVERSE REACTIONS
The following adverse events, occurring in less than 2% of patients, were reported in association with the use of IOPIDINE 1% Ophthalmic Solution in laser surgery: ocular injection, upper lid elevation, irregular heart rate, nasal decongestion, ocular inflammation, conjunctival blanching, and mydriasis.
DOSAGE AND ADMINISTRATION
Instill one drop of IOPIDINE® 1% in the operative eye one hour before anterior segment laser surgery. Instill a second drop immediately after the procedure. Use a new single-use container for each drop and discard after use.
You are encouraged to report suspected adverse reactions to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.

ILEVRO®
(nepafenac ophthalmic suspension) 0.3%
More Information
Indications and Usage
ILEVRO® (nepafenac ophthalmic suspension) 0.3% is a nonsteroidal, anti-inflammatory prodrug indicated for the treatment of pain and inflammation associated with cataract surgery.
Dosage and Administration
One drop of ILEVRO® 0.3% should be applied to the affected eye one-time-daily beginning 1 day prior to cataract surgery, continued on the day of surgery and through the first 2 weeks of the postoperative period. An additional drop should be administered 30 to 120 minutes prior to surgery.
Important Safety information
Contraindications
ILEVRO® 0.3% is contraindicated in patients with previously demonstrated hypersensitivity to any of the ingredients in the formula or to other nonsteroidal anti-inflammatory drugs (NSAIDs).
Warnings and Precautions
- Increased Bleeding Time – With some NSAIDs, including ILEVRO® 0.3%, there exists the potential for increased bleeding time. Ocularly applied NSAIDs may cause increased bleeding of ocular tissues (including hyphema) in conjunction with ocular surgery.
- Delayed Healing – Topical NSAIDs, including ILEVRO® 0.3%, may slow or delay healing. Concomitant use of topical NSAIDs and topical steroids may increase the potential for healing problems.
- Corneal Effects – Use of topical NSAIDs may result in keratitis. In some patients, continued use of topical NSAIDs may result in epithelial breakdown, corneal thinning, corneal erosion, corneal ulceration, or corneal perforation. These events may be sight threatening. Patients with evidence of corneal epithelial breakdown should immediately discontinue use of topical NSAIDs, including ILEVRO® 0.3%, and should be closely monitored for corneal health.
Patients with complicated ocular surgeries, corneal denervation, corneal epithelial defects, diabetes mellitus, ocular surface diseases (e.g., dry eye syndrome), rheumatoid arthritis, or repeat ocular surgeries within a short period of time may be at increased risk for corneal adverse events, which may become sight threatening. Topical NSAIDs should be used with caution in these patients.
Use more than 1 day prior to surgery or use beyond 14 days post-surgery may increase patient risk and severity of corneal adverse events.
- Contact Lens Wear – ILEVRO® 0.3% should not be administered while using contact lenses.
Adverse Reactions
The most frequently reported ocular adverse reactions following cataract surgery occurring in approximately 5% to 10% of patients were capsular opacity, decreased visual acuity, foreign body sensation, increased intraocular pressure, and sticky sensation.

More Information
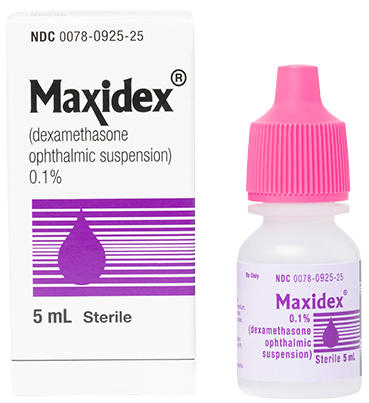
MAXIDEX®
(dexamethasone ophthalmic suspension) 0.1%
More Information
Indications and Usage
Steroid responsive inflammatory conditions of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the globe, such as allergic conjunctivitis, acne rosacea, superficial punctate keratitis, herpes zoster keratitis, iritis, cyclitis, selected infective conjunctivitides when the inherent hazard of steroid use is accepted to obtain an advisable diminution in edema and inflammation; corneal injury from chemical, radiation, or thermal burns, or penetration of foreign bodies.
Important Safety information
CONTRAINDICATIONS
Contraindicated in acute, untreated bacterial infections; mycobacterial ocular infections; epithelial herpes simplex (dendritic keratitis); vaccinia, varicella, and most other viral diseases of the cornea and conjunctiva; fungal disease of ocular structures; and in those persons who have shown hypersensitivity to any component of this preparation.
WARNINGS AND PRECAUTIONS
Prolonged use may result in ocular hypertension and/or glaucoma, with damage to the optic nerve, defects in visual acuity and fields of vision, and posterior subcapsular cataract formation. Prolonged use may suppress the host response and thus increase the hazard of secondary ocular infections. In acute purulent conditions or parasitic infections of the eye, corticosteroids may mask infection or enhance existing infection. In those diseases causing thinning of the cornea or sclera, perforations have been known to occur with the use of topical corticosteroids. If these products are used for 10 days or longer, intraocular pressure (IOP) should be routinely monitored even though it may be difficult in children and uncooperative patients. Employment of corticosteroid medication in the treatment of herpes simplex other than epithelial herpes simplex keratitis, in which it is contraindicated, requires great caution; periodic slit-lamp microscopy is essential.
ADVERSE REACTIONS
Glaucoma with optic nerve damage, visual acuity and field defects; cataract formation; secondary ocular infection following suppression of host response; and perforation of the globe may occur.
DOSAGE AND ADMINISTRATION
One or two drops topically in the conjunctival sac(s). In severe disease, drops may be used hourly, being tapered to discontinuation as the inflammation subsides. In mild disease, drops may be used up to four to six times daily.
You are encouraged to report suspected adverse reactions to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.

MAXITROL®
(neomycin and polymixin B sulfates and dexamthasone ophthalmic suspension)
More Information
Indications and Usage
For steroid-responsive inflammatory ocular conditions for which a corticosteroid is indicated and where bacterial infection or a risk of bacterial ocular infection exists.
Ocular steroids are indicated in inflammatory conditions of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the globe where the inherent risk of steroid use in certain infective conjunctivitides is accepted to obtain a diminution in edema and inflammation. They are also indicated in chronic anterior uveitis and corneal injury from chemical, radiation or thermal burns; or penetration of foreign bodies.
The use of a combination drug with an anti-infective component is indicated where the risk of infection is high or where there is an expectation that potentially dangerous numbers of bacteria will be present in the eye.
The particular anti-infective drug in this product is active against the following common bacterial eye pathogens: Staphylococcus aureus, Escherichia coli, Haemophilus influenzae, Klebsiella/Enterobacter species, Neisseria species, and Pseudomonas aeruginosa.
This product does not provide adequate coverage against: Serratia marcescens and Streptococci, including Streptococcus pneumoniae.
Important Safety information
CONTRAINDICATIONS
Maxitrol is contraindicated in patients with epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, varicella, and many other viral infections of the cornea and conjunctiva, mycobacterial infections of the eye, fungal diseases of ocular structures, as well as hypersensitivity to any component of the medication.
WARNINGS
Maxitrol should not be injected or intraocularly administered.
Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye. Prolonged use may result in glaucoma, with damage to the optic nerve, defects in visual acuity and fields of vision, and posterior subcapsular cataract formation. Prolonged use may suppress the host response and thus increase the hazard of secondary ocular infection.
If this product is used for 10 days or longer, intraocular pressure should be routinely monitored.
Neomycin sulfate may cause cutaneous sensitization. Sensitivity to topically administered aminoglycosides, such as neomycin, may occur in some patients. If hypersensitivity develops, treatment should be discontinued. Cross hypersensitivity to other aminoglycosides can occur, and the possibility that patients who become sensitized to topical neomycin may also be sensitive to other topical and/or systemic aminoglycosides should be considered.
PRECAUTIONS
If inflammation or pain persists longer than 48 hours or becomes aggravated, the patient should discontinue use and consult a physician. Patients should be advised that their vision may be temporarily blurred following dosing with Maxitrol.
ADVERSE REACTIONS
Reactions occurring most often from the presence of the anti-infective ingredient are allergic sensitizations. The reactions due to the steroid component are: elevation of intraocular pressure (IOP) with possible development of glaucoma, and infrequent optic nerve damage; posterior subcapsular cataract formation; and delayed wound healing.
You are encouraged to report suspected adverse reactions to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.

NATACYN®
(natamycin ophthalmic suspension) 5%
More Information
Indications and Usage
NATACYN® (natamycin ophthalmic suspension) 5% is indicated for the treatment of fungal blepharitis, conjunctivitis, and keratitis caused by susceptible organisms including Fusarium solani keratitis. As in other forms of suppurative keratitis, initial and sustained therapy of fungal keratitis should be determined by the clinical diagnosis, laboratory diagnosis by smear and culture of corneal scrapings and drug response. Whenever possible the in vitro activity of natamycin against the responsible fungus should be determined. The effectiveness of natamycin as a single agent in fungal endophthalmitis has not been established.
Important Safety information
CONTRAINDICATIONS
NATACYN® (natamycin ophthalmic suspension) 5% is contraindicated in individuals with a history of hypersensitivity to any of its components.
PRECAUTIONS
- General: FOR TOPICAL OPHTHALMIC USE ONLY — NOT FOR INJECTION. Failure of improvement of keratitis following 7-10 days of administration of the drug suggests that the infection may be caused by a microorganism not susceptible to natamycin. Continuation of therapy should be based on clinical re-evaluation and additional laboratory studies.
Continuation of therapy should be based on clinical re-evaluation and additional laboratory studies.
Adherence of the suspension to areas of epithelial ulceration or retention of the suspension in the fornices occurs regularly.
Use only if the container is undamaged. - Information for Patients: Do not touch dropper tip to any surface, as this may contaminate the suspension. Patients should be advised not to wear contact lenses if they have signs and symptoms of fungal blepharitis, conjunctivitis, and keratitis.
- Pregnancy: NATACYN® (natamycin ophthalmic suspension) 5% should be given to a pregnant woman only if benefits outweigh risks.
- Nursing Mothers: Caution should be exercised when natamycin is administered to a nursing woman.
- Pediatric Use: Safety and effectiveness in pediatric patients have not been established.
ADVERSE REACTIONS
The following events have been identified during post-marketing use of NATACYN® in clinical practice. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The events, which have been chosen for inclusion due to their seriousness, frequency of reporting, possible causal connection to NATACYN®, or a combination of these factors include: allergic reaction, change in vision, chest pain, corneal opacity, dyspnea, eye discomfort, eye edema, eye hyperemia, eye irritation, eye pain, foreign body sensation, paresthesia, and tearing.


More Information
NEVANAC®
(nepafenac ophthalmic suspension) 0.1%
More Information
Indications and Usage
NEVANAC ophthalmic suspension is a nonsteroidal, anti-inflammatory prodrug indicated for the treatment of pain and inflammation associated with cataract surgery.
This product does not provide adequate coverage against: Serratia marcescens and Streptococci, including Streptococcus pneumoniae.
Important Safety information
CONTRAINDICATIONS
Hypersensitivity to any of the ingredients in the formula or to other NSAIDS.
WARNINGS & PRECAUTIONS
- Increased Bleeding Time – With some nonsteroidal anti-inflammatory drugs including NEVANAC®, there exists the potential for increased bleeding time due to interference with thrombocyte aggregation. There have been reports that ocularly applied nonsteroidal anti-inflammatory drugs may cause increased bleeding of ocular tissues (including hyphemas) in conjunction with ocular surgery. It is recommended that NEVANAC® ophthalmic suspension be used with caution in patients with known bleeding tendencies or who are receiving other medications which may prolong bleeding time.
- Delayed Healing – Topical nonsteroidal anti-inflammatory drugs (NSAIDs) including NEVANAC®, may slow or delay healing. Topical corticosteroids are also known to slow or delay healing. Concomitant use of topical NSAIDs and topical steroids may increase the potential for healing problems.
Corneal Effects – Use of topical NSAIDs may result in keratitis. In some susceptible patients, continued use of topical NSAIDs may result in epithelial breakdown, corneal thinning, corneal erosion, corneal ulceration or corneal perforation. These events may be sight threatening. Patients with evidence of corneal epithelial breakdown should immediately discontinue use of topical NSAIDs including NEVANAC® and should be closely monitored for corneal health.
Postmarketing experience with topical NSAIDs suggests that patients with complicated ocular surgeries, corneal denervation, corneal epithelial defects, diabetes mellitus, ocular surface diseases (e.g., dry eye syndrome),rheumatoid arthritis, or repeat ocular surgeries within a short period of time may be at increased risk for corneal adverse events which may become sight threatening. Topical NSAIDs should be used with caution in these patients.
Postmarketing experience with topical NSAIDs also suggests that use more than 1 day prior to surgery or use beyond 14 days post surgery may increase patient risk and severity of corneal adverse events.
- Contact Lens Wear – NEVANAC should not be administered while using contact lenses.
ADVERSE REACTIONS
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to the rates in the clinical studies of another drug and may not reflect the rates observed in practice.
DOSING AND ADMINISTRATION
One drop of NEVANAC should be applied to the affected eye three-times-daily beginning 1 day prior to cataract surgery, continued on the day of surgery and through the first 2 weeks of the postoperative period.
You are encouraged to report suspected adverse reactions to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.

TOBRADEX® ST
(tobramycin/dexamethasone ophthalmic suspension) 0.3%/0.05%
More Information
Indications and Usage
TOBRADEX® ST (tobramycin and dexamethasone ophthalmic suspension) 0.3%/0.05% is a topical antibiotic and corticosteroid combination for steroid-responsive inflammatory ocular conditions for which a corticosteroid is indicated and where superficial bacterial ocular infection or a risk of bacterial ocular infection exists.
Important Safety information
CONTRAINDICATIONS
Most viral diseases of the cornea and conjunctiva, including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, varicella, and also in mycobacterial infection of the eye and fungal disease of ocular structures. Hypersensitivity to any components of the medication.
WARNINGS & PRECAUTIONS
- Intraocular Pressure (IOP) Increase - Prolonged use may result in glaucoma with damage to the optic nerve, and defects in visual acuity and fields of vision. IOP should be monitored.
- Aminoglycoside Sensitivity - Sensitivity to topically applied aminoglycosides may occur.
- Cataracts - Posterior subcapsular cataract formation may occur.
- Delayed Healing - May delay healing and increase the incidence of bleb formation. Perforations of the cornea or sclera have occurred. Slit lamp biomicroscopy and fluorescein staining should be conducted.
- Bacterial Infections - May suppress host response and increase secondary ocular infections. In acute purulent conditions, steroids may mask infection or enhance existing infection. If signs and symptoms fail to improve after 2 days, then the patient should be re-evaluated.
- Viral Infections - Use with a history of herpes simplex requires great caution. The course and severity of many viral infections of the eye (including herpes simplex) may be exacerbated.
- Fungal Infections - Fungal infections of the cornea may occur and should be considered in any persistent corneal ulceration.
- Vision Blurred - Vision may be temporarily blurred following dosing with TOBRADEX® ST. Care should be exercised in operating machinery or driving a motor vehicle.
- Risk of Contamination - Do not touch the dropper tip of the bottle to any surface, as this may contaminate the contents.
- Contact Lens Use - TOBRADEX® ST contains benzalkonium chloride, an antimicrobial preservative, which may be absorbed by soft contact lenses. Contact lenses should not be worn during the use of TOBRADEX® ST.
ADVERSE REACTIONS
The most frequent adverse reactions (<4%) to topical ocular tobramycin are hypersensitivity and localized ocular toxicity, including eye pain, eyelid pruritus, eyelid edema, and conjunctival hyperemia.
The reactions due to the steroid component are increased IOP with possible damage to optic nerve and development of glaucoma, subcapsular cataract, and impaired healing.
The development of secondary infection has occurred. Fungal infections of the cornea may occur. Secondary bacterial ocular infection following suppression of host responses also occurs.
Non-ocular adverse events (0.5% to 1%) included headache and increased blood pressure.
The following additional adverse reactions have been reported with the individual components below:
- Aminoglycosides: Neurotoxicity, ototoxicity, and nephrotoxicity have occurred in patients receiving systemic aminoglycoside therapy. Aminoglycosides may aggravate muscle weakness in patients with known or suspected neuromuscular disorders, such as myasthenia gravis or Parkinson’s disease, because of their potential effect on neuromuscular function.
- Dexamethasone: Cushing’s syndrome and adrenal suppression may occur after the use of dexamethasone in excess of the listed dosing instructions in predisposed patients, including children and patients treated with CYP3A4 inhibitors.
POSTMARKETING EXPERIENCE
The following adverse reactions have been identified during post approval use of TOBRADEX® ST. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Additional adverse reactions identified from post approval use include anaphylactic reaction and erythema multiforme.

TRIESENCE®
(triamcinolone acetonide injectable suspension) 40 mg/mL
More Information
Indications and Usage
TRIESENCE® Suspension is indicated for:
- Treatment of the following ophthalmic diseases: sympathetic ophthalmia, temporal arteritis, uveitis, and ocular inflammatory conditions unresponsive to topical corticosteroids.
- Visualization during vitrectomy.
Important Safety information
CONTRAINDICATIONS
- TRIESENCE® Suspension is contraindicated in patients with systemic fungal infections.
- TRIESENCE® Suspension is also contraindicated in patients with hypersensitivity to corticosteroids or any component of TRIESENCE® Suspension. Rare instances of anaphylactoid reactions have occurred in patients receiving corticosteroid therapy.
WARNINGS AND PRECAUTIONS
- TRIESENCE® is a suspension; it should not be administered intravenously.
- Ophthalmic effects: May include cataracts, infections, and glaucoma. Monitor intraocular pressure.
- Hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing’s syndrome and hyperglycemia: Monitor patients for these conditions and taper doses gradually.
- Infections: Increased susceptibility to new infection and increased risk of exacerbation, dissemination, or reactivation of latent infection.
- Elevated blood pressure, salt and water retention, and hypokalemia: Monitor blood pressure and sodium, potassium serum levels.
- GI perforation: Increased risk in patients with certain GI disorders.
- Behavioral and mood disturbances: May include euphoria, insomnia, mood swings, personality changes, severe depression, and psychosis.
- Decreases in bone density: Monitor bone density in patients receiving long term corticosteroid therapy.
- Live or live attenuated vaccines: Do not administer to patients receiving immunosuppressive doses of corticosteroids.
- Negative effects on growth and development: Monitor pediatric patients on long-term corticosteroid therapy.
- Use in pregnancy: Fetal harm can occur with first trimester use.
- Weight gain: May cause increased appetite.
ADVERSE REACTIONS
- Based on a review of the available literature, the most commonly reported adverse events following ocular administration of triamcinolone acetonide were elevated intraocular pressure and cataract progression. These events have been reported to occur in 20-60% of patients.
- Less common reactions occurring in up to 2% of patients include: endophthalmitis (infectious and non-infectious), hypopyon, injection site reactions (described as blurring and transient discomfort), glaucoma, vitreous floaters, detachment of retinal pigment epithelium, optic disc vascular disorder, eye inflammation, conjunctival hemorrhage and visual acuity reduced. Cases of exophthalmos have also been reported.
DRUG INTERACTIONS
- Anticoagulant Agents –Corticosteroids may enhance or diminish the anticoagulant effect of anticoagulant agents. Coagulation indices should be monitored.
- Antidiabetic Agents –Corticosteroids may increase blood glucose concentrations. Dose adjustments of antidiabetic agents may be required.
- CYP 3A4 Inducers and Inhibitors –CYP 3A4 inducers and inhibitors may respectively increase or decrease clearance of corticosteroids, necessitating dose adjustment.
- NSAIDs –Concomitant use of NSAIDS, including aspirin and salicylates, with a corticosteroid may increase the risk of GI side effects.

VERKAZIA®
(cyclosporine ophthalmic emulsion) 0.1%
More Information
Indications and Usage
Verkazia® (cyclosporine ophthalmic emulsion) 0.1% is a calcineurin inhibitor immunosuppressant indicated for the treatment of vernal keratoconjunctivitis in children and adults.
Important Safety information
WARNINGS AND PRECAUTIONS
Potential for eye injury and contamination: To avoid the potential for eye injury and contamination, advise patient not to touch the vial tip to the eye or other surfaces.
ADVERSE REACTIONS
The most common adverse reactions reported in greater than 5% of patients were eye pain (12%) and eye pruritus (8%), which were usually transitory and occurred during instillation.
You are encouraged to report adverse reactions of prescription drugs to FDA at 1-800-FDA-1088. You may also report side effects to Harrow at 1-833-4HARROW (1-833-442-7769).

VEVYE®
(cyclosporine ophthalmic solution) 0.1%
More Information
Indications and Usage
VEVYE® (cyclosporine ophthalmic solution) 0.1% is indicated for the treatment of the signs and symptoms of dry eye disease.
Important Safety information
WARNINGS AND PRECAUTIONS
- Potential for Eye Injury and Contamination – To avoid the potential for eye injury and/or contamination, patients should not touch the bottle tip to the eye or other surfaces.
- Use with Contact Lenses – VEVYE® should not be administered while wearing contact lenses. If contact lenses are worn, they should be removed prior to administration of the solution. Lenses may be reinserted 15 minutes following the administration of VEVYE®.
ADVERSE REACTIONS
In clinical trials with 738 subjects receiving at least 1 dose of VEVYE®, the most common adverse reactions were instillation site reactions (8%) and temporary decreases in visual acuity (3%).
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.

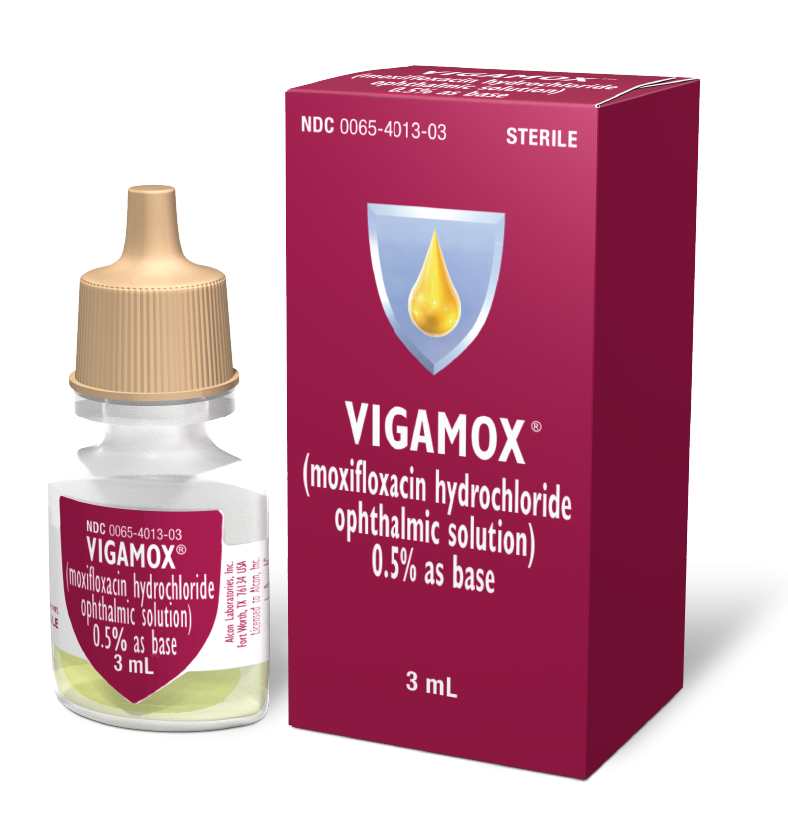
VIGAMOX®
(moxifloxacin HCI ophthalmic solution) 0.5%
More Information
Indications and Usage
VIGAMOX® (moxifloxacin HCI ophthalmic solution) 0.5% as base is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms: Corynebacterium species,* Micrococcus luteus,* Staphylococcus aureus, S. epidermidis, S. haemolyticus, S. hominis, S. warneri, * Streptococcus pneumoniae, S. viridans group, Acinetobacter lwoffii,* Haemophilus influenzae, H. parainfluenzae,* Chlamydia trachomatis (*efficacy for this organism was studied in fewer than 10 infections).
Important Safety information
WARNINGS AND PRECAUTIONS
- Hypersensitivity Reactions – In patients receiving systemically administered quinolones, including moxifloxacin, serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported, some following the first dose. If an allergic reaction to moxifloxacin occurs, discontinue use of the drug.
- Prolonged Use – Prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. If superinfection occurs, discontinue use and institute alternative therapy.
- Avoidance of Contact Lens Wear – Patients should be advised not to wear contact lenses if they have signs or symptoms of bacterial conjunctivitis.
ADVERSE REACTIONS
In VIGAMOX® Solution clinical trials, the most frequently reported ocular adverse events were conjunctivitis, decreased visual acuity, dry eye, keratitis, ocular discomfort, ocular hyperemia, ocular pain, ocular pruritus, subconjunctival hemorrhage, and tearing. These events occurred in approximately 1–6% of patients. Nonocular adverse events reported at a rate of 1–4% were fever, increased cough, infection, otitis media, pharyngitis, rash, and rhinitis.
You are encouraged to report suspected adverse reactions to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.

ZERVIATE®
cetirizine ophthalmic solution, 0.24%
More Information
Indications and Usage
ZERVIATE® (cetirizine ophthalmic solution) 0.24% is a histamine-1 (H1) receptor antagonist indicated for the treatment of ocular itching associated with allergic conjunctivitis.
Important Safety information
WARNINGS AND PRECAUTIONS
Contamination of Tip and Solution: As with any eye drop, care should be taken not to touch the eyelids or surrounding areas with the dropper tip of the bottle or tip of the single-use container to avoid injury to the eye and to prevent contaminating the tip and solution. Keep the multi-dose bottle closed when not in use. Discard the single-use container after using in each eye.
Contact Lens Wear: Patients should be advised not to wear a contact lens if their eye is red. ZERVIATE should not be instilled while wearing contact lenses. Remove contact lenses prior to instillation of ZERVIATE. The preservative in ZERVIATE, benzalkonium chloride, may be absorbed by soft contact lenses. Lenses may be reinserted 10 minutes following administration of ZERVIATE.
ADVERSE REACTIONS
The most commonly reported adverse reactions occurred in approximately 1%–7% of patients treated with either ZERVIATE or vehicle. These reactions were ocular hyperemia, instillation site pain, and visual acuity reduced.
Available from ImprimisRx:
Harrow owns an FDA-Registered 503B Outsourcing Facility and a patient-specific 503A pharmacy.


Product Information
For information about a Harrow product:
- Visit our Products page.
- Click "Learn More" beneath the product you're interested in.
- On the product’s page, look for a “Contact Us” link — this is often the fastest way to get product-specific information.
Contact one of our Customer Support teams for questions, including:
- Product quality complaints
- Adverse event reporting
- Requests for medical information
- General inquiries
FDA-approved drug products:
Phone: 833-442-7769 (833-4HARROW)
Email: HE-QA@harrowinc.com
Compounded products:
Phone: 844-446-6979
Email: info@ImprimisRx.com
![]()
Returned Goods Policy
To ensure safe and compliant product handling, all Harrow returns must be authorized in advance. Unauthorized returns may be destroyed without credit or replacement. Eligible products, purchased directly from Harrow or an authorized distributor, must be returned in original, unopened packaging within the allowed return window.
For full details on return eligibility, timing, documentation, and shipping instructions, please refer to our Returned Goods Policy.